Three-Level Anterior Cervical Discectomy and Fusion With or Without an Investigational Posterior Stabilization System Assessed Through 24 Months A Multicenter Randomized Controlled Trial
Spine 50(17):p 1161-1170, September 1, 2025. | DOI: 10.1097/BRS.0000000000005387
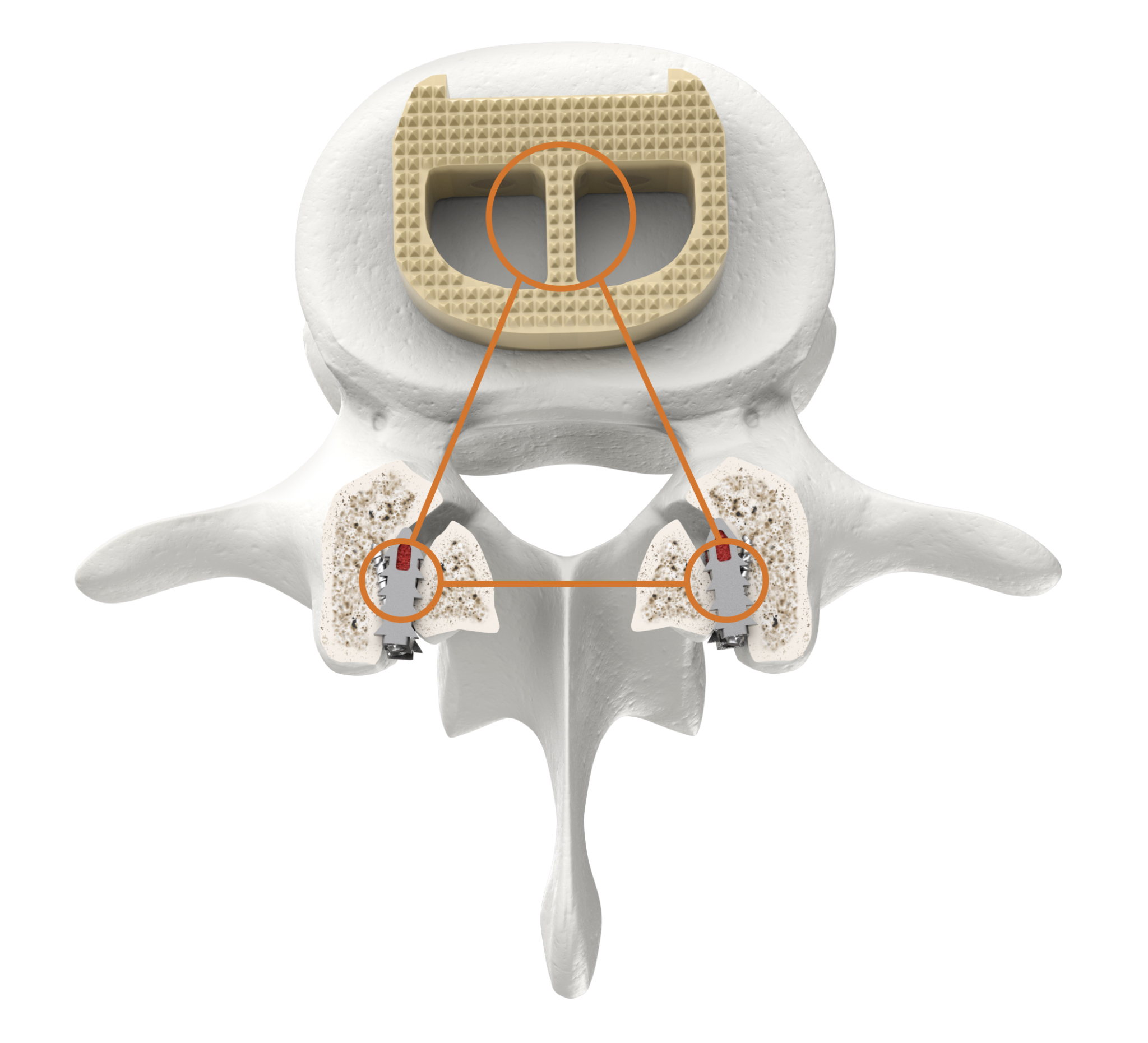
Anterior fusion alone is not enough for certain patients to reliably achieve fusion.1
CORUS™ offers the potential for improved fusion rates with little added morbidity and no change on hospital length of stay.1
A spinal motion segment is not fully fused unless all joints are fused.